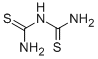
CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Dithiobiuret is a crystalline solid. Used as a plasticizer, as a rubber accelerator, and as an intermediate in manufacturing of pesticides. (EPA, 1998) |
|---|---|
| Color/Form | MONOCLINIC OR TRICLINIC CRYSTALS |
| Melting Point | 358 °F decomposes (EPA, 1998) |
| Solubility | @ 27 °C: 0.27 G/100 ML WATER, 2.2 G/100 G ETHANOL, 16 G/100 G ACETONE, ABOUT 34 G/100 G CELLOSOLVE; IN BOILING WATER= ABOUT 8%; IN 1%SODIUM HYDROXIDE= 3.6 G/100 G; IN 5% SODIUM HYDROXIDE= 16 G/100 G; IN 10% SODIUM HYDROXIDE= 29 G/100 G; SOL IN ALKALIES WITH FORMATION OF WATER-SOLUBLE SALTS. |
| Density | 1.522 at 86 °F (EPA, 1998) - Denser than water; will sink |
| Decomposition | DECOMPOSES @ 181 °C |
| pH | SATURATED AQ SOLN @ 30 °C: 5.8 |
| Chemical Classes | Nitrogen Compounds -> Thiourea Compounds |
SAFETY INFORMATION
| Signal word | Danger |
|---|---|
| Pictogram(s) |
 Skull and Crossbones Acute Toxicity GHS06 |
| Precautionary Statement Codes |
P260:Do not breathe dust/fume/gas/mist/vapours/spray. P262:Do not get in eyes, on skin, or on clothing. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P280:Wear protective gloves/protective clothing/eye protection/face protection. |
COMPUTED DESCRIPTORS
| Molecular Weight | 135.22 g/mol |
|---|---|
| XLogP3 | -0.6 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 134.99248952 g/mol |
| Monoisotopic Mass | 134.99248952 g/mol |
| Topological Polar Surface Area | 128 Ų |
| Heavy Atom Count | 7 |
| Formal Charge | 0 |
| Complexity | 87.1 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Dithiobiuret is a crystalline solid. Used as a plasticizer, as a rubber accelerator, and as an intermediate in manufacturing of pesticides. (EPA, 1998)