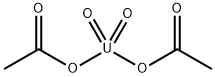
CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Uranyl acetate appears as yellow crystals with a slight odor of vinegar. Density 2.89 g / cm3. |
|---|---|
| Odor | Vinegar-like odor |
| Density | 2.89 at 68 °F (USCG, 1999) - Denser than water; will sink |
| Decomposition | When heated to decomposition it emits toxic fumes of uranium. |
| Other Experimental Properties | Yellow crystalline powder; slight acetic odor; freely soluble in water acidulated with acetic acid; soluble in 10 parts of water, usually incompletely, due to the presence of the basic salt /Dihydrate/ |
| Chemical Classes | Physical/Radiation -> Radioactive Compounds |
COMPUTED DESCRIPTORS
| Molecular Weight | 390.13 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 0 |
| Exact Mass | 390.08287 g/mol |
| Monoisotopic Mass | 390.08287 g/mol |
| Topological Polar Surface Area | 109 Ų |
| Heavy Atom Count | 11 |
| Formal Charge | 0 |
| Complexity | 129 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 3 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Uranyl acetate appears as yellow crystals with a slight odor of vinegar. Density 2.89 g / cm3.