SAFETY INFORMATION
| Signal word | Warning |
|---|---|
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
COMPUTED DESCRIPTORS
| Molecular Weight | 406.4 g/mol |
|---|---|
| XLogP3 | 2.4 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 8 |
| Exact Mass | 406.14526146 g/mol |
| Monoisotopic Mass | 406.14526146 g/mol |
| Topological Polar Surface Area | 108 Ų |
| Heavy Atom Count | 29 |
| Formal Charge | 0 |
| Complexity | 591 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
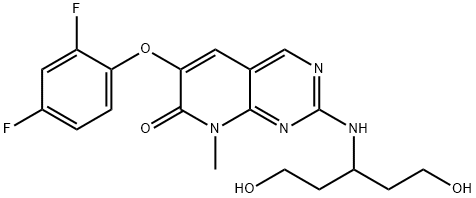
Pamapimod is a member of the class of pyridopyrimidines that is 8-methylpyrido[2,3-d]pyrimidin-7(8H)-one carrying additional (1,5-dihydroxypentan-3-yl)amino and 2,4-difluorophenoxy substituents at positions 2 and 6 respectively. It is a potent inhibitor of MAPK and is used for treatment of rheumatoid arthritis. It has a role as an EC 2.7.11.24 (mitogen-activated protein kinase) inhibitor and an antirheumatic drug. It is a pyridopyrimidine, an aromatic ether, a difluorobenzene, an aromatic amine, a secondary amino compound, a primary alcohol and a diol.