CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Solid |
|---|---|
| Color/Form | Off-white to pale-yellow solid powder |
| Solubility | Soluble in organic solvent/water mixtures, and buffered aqueous solvents. Lenalidomide is more soluble in organic solvents and low pH solutions. Solubility was significantly lower in less acidic buffers, ranging from about 0.4 to 0.5 mg/mL. |
| LogP | -0.4 |
| Optical Rotation | Has an asymmetric carbon atom and can exist as the optically active forms S(-) and R(+), and is produced as a reacemic mixture with a net optical rotation of zero |
COMPUTED DESCRIPTORS
| Molecular Weight | 259.26 g/mol |
|---|---|
| XLogP3 | -0.5 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 1 |
| Exact Mass | 259.09569129 g/mol |
| Monoisotopic Mass | 259.09569129 g/mol |
| Topological Polar Surface Area | 92.5 Ų |
| Heavy Atom Count | 19 |
| Formal Charge | 0 |
| Complexity | 437 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
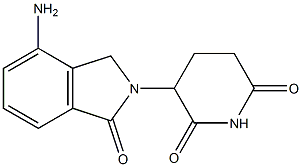
Lenalidomide is a dicarboximide that consists of 1-oxoisoindoline bearing an amino substituent at position 4 and a 2,6-dioxopiperidin-3-yl group at position 2. Inhibits the secretion of TNF-alpha. It has a role as an angiogenesis inhibitor, an antineoplastic agent and an immunomodulator. It is a member of isoindoles, a dicarboximide, a member of piperidones and an aromatic amine.