305335-31-3
Product Name:
MLN-4760
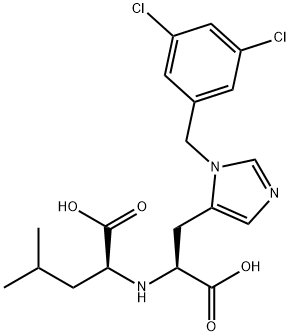
Formula:
C19H23Cl2N3O4
Synonyms:
;ACE2 Inhibitor, MLN-4760
Inquiry
SAFETY INFORMATION
| Signal word | Warning |
|---|---|
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H312:Acute toxicity,dermal H332:Acute toxicity,inhalation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P270:Do not eat, drink or smoke when using this product. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P312:Call a POISON CENTER or doctor/physician if you feel unwell. P330:Rinse mouth. P362:Take off contaminated clothing and wash before reuse. P302+P352:IF ON SKIN: wash with plenty of soap and water. P304+P340:IF INHALED: Remove victim to fresh air and Keep at rest in a position comfortable for breathing. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P403+P233:Store in a well-ventilated place. Keep container tightly closed. P501:Dispose of contents/container to..… |
COMPUTED DESCRIPTORS
| Molecular Weight | 428.3 g/mol |
|---|---|
| XLogP3 | 1.4 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 10 |
| Exact Mass | 427.1065616 g/mol |
| Monoisotopic Mass | 427.1065616 g/mol |
| Topological Polar Surface Area | 104 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 527 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
MLN-4760 is a L-histidine derivative that is L-histidine in which a hydrogen of the primary amino group is substituted by a (1S)-1-carboxy-3-methylbutyl group and the ring NH group is substituted by a 3,5-dichlorobenzyl group. It is a potent and selective human angiotensin-converting enzyme 2 (ACE2) inhibitor (IC50 = 0.44 nM) which was in clinical development for the treatment of ulcerative colitis. It has a role as an anti-inflammatory agent and an EC 3.4.17.23 (angiotensin-converting enzyme 2) inhibitor. It is a L-histidine derivative, a dichlorobenzene and a L-leucine derivative.
