CHEMICAL AND PHYSICAL PROPERTIES
| Collision Cross Section | 189.99 Ų [M+H]+ [CCS Type: TW] |
|---|
COMPUTED DESCRIPTORS
| Molecular Weight | 373.4 g/mol |
|---|---|
| XLogP3 | 3.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 6 |
| Exact Mass | 373.18892296 g/mol |
| Monoisotopic Mass | 373.18892296 g/mol |
| Topological Polar Surface Area | 73.9 Ų |
| Heavy Atom Count | 27 |
| Formal Charge | 0 |
| Complexity | 606 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
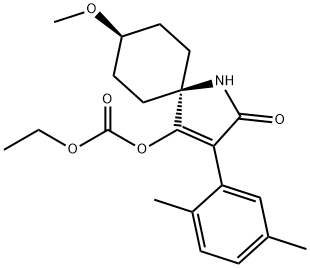
Spirotetramat is an azaspiro compound that is methoxycyclohexane which is fused at position 4 to the 5-position of a 1,5-dihydro-2H-pyrrol-2-one that is substituted at positions 3 and 4 by 2,5-dimethylphenyl and (ethoxycarbonyl)oxy groups, respectively (the cis isomer). It is a proinsecticide (via hydrolysis of the ethyl carbonate group to give the corresponding 4-hydroxypyrrol-2-one, "spirotetramat-enol") and is used for the control of a wide range of sucking insects on fruit and potato crops. It has a role as a proinsecticide, an EC 6.4.1.2 (acetyl-CoA carboxylase) inhibitor and an agrochemical. It is a member of benzenes, an azaspiro compound, a carbonate ester, a gamma-lactam and a member of pyrroles.