CHEMICAL AND PHYSICAL PROPERTIES
| Kovats Retention Index | 1628 1627 1627 1635 1628 1626 1629 1620 1629 1644 1645 1643 1630 1625 1636 1628 1615 1627 1627 1627 1633 1611 1623 1641 1627 1654 1621 1630 1622 1622 1630 1638 1649 1606 1619 1608 1639 1659 1651 1625 1631 1620 1630 1633 1632 1630 1622 1638 1635 1633 1635 1620 1637 1633 1635 1633 1637 1642 1630 1615 1640 1629 1624 1640 1663 1638 1633 1631 1643 1634 1638 1610 1642 1634 1640 1643 1627 1632 1633 1635 1636 1635 1597 1607 1625.3 1620 1637 1630.9 1641.9 1627 1632 1641 1636 1644 1615 1610 1634 1635 1636 1633 1628 1634 1650 1616 1636 1629 1645 1650 1616 1630 1632 1635 1635 1624 1621 1639 1626 1631 1635 |
|---|
COMPUTED DESCRIPTORS
| Molecular Weight | 222.37 g/mol |
|---|---|
| XLogP3 | 3.3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 1 |
| Exact Mass | 222.198365449 g/mol |
| Monoisotopic Mass | 222.198365449 g/mol |
| Topological Polar Surface Area | 20.2 Ų |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 292 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
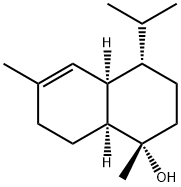
(-)-Tau-muurolol is a cadinane sesquiterpenoid that consists of 4-isopropyl-1,6-dimethyl-1,2,3,4,4a,7,8,8a-octahydronaphthalene having a hydroxy substituent at position 1 and (1S,4S,4aR,8aS)-configuration. It has a role as a plant metabolite, a fungicide, a volatile oil component, a marine metabolite and a bacterial metabolite. It is a cadinane sesquiterpenoid, a carbobicyclic compound, a tertiary alcohol and a member of octahydronaphthalenes. It is an enantiomer of a (+)-Tau-muurolol.