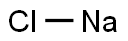CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Dry Powder; Dry Powder, Liquid; Dry Powder, Other Solid; Dry Powder, Water or Solvent Wet Solid; Liquid; Liquid, Other Solid; NKRA; Other Solid; Pellets or Large Crystals; Pellets or Large Crystals, Liquid; Water or Solvent Wet Solid |
|---|---|
| Color/Form | Colorless, transparent crystals or white, crystalline powder |
| Taste | SRP: SALTY |
| Boiling Point | 1465 °C |
| Melting Point | 800.7 °C |
| Solubility | 36.0 g/100 g of water at 25 °C |
| Density | 2.17 at 25 °C/4 °C |
| Vapor Pressure | VP: 1 mm Hg at 865 °C |
| Stability/Shelf Life | Stable under recommended storage conditions. |
| Decomposition | When heated to decomposition it emits toxic fumes of /hydrochloric acid and disodium oxide/. |
| Viscosity | Viscosity of saturated aqueous solution = 1.93 mPa-s |
| Corrosivity | Sodium chloride solutions are corrosive to base metals |
| pH | pH = 6.7 to 7.3; its aqueous solution is neutral |
| Surface Tension | 110 mN/m at 850 °C /Molten sodium chloride/ |
| Refractive Index | Index of refraction: 1.5442 /Table/ |
| Other Experimental Properties | A 23% aqueous solution of sodium chloride freezes at -20.5 °C. ... Density of saturated aqueous solution at 25 °C is 1.202 ... Begins to volatilize at a little above its melting point. ... Its solubility is decreased by hydrogen chloride. |
| Chemical Classes | Metals -> Metals, Inorganic Compounds |
COMPUTED DESCRIPTORS
| Molecular Weight | 58.44 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 0 |
| Exact Mass | 57.9586220 g/mol |
| Monoisotopic Mass | 57.9586220 g/mol |
| Topological Polar Surface Area | 0 Ų |
| Heavy Atom Count | 2 |
| Formal Charge | 0 |
| Complexity | 2 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 2 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Sodium chloride is an inorganic chloride salt having sodium(1+) as the counterion. It has a role as an emetic and a flame retardant. It is an inorganic chloride and an inorganic sodium salt.