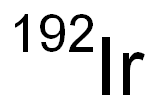CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | A hard and brittle metal; It is about twice as dense as lead; Some of the salts are highly colored; [Argonne] |
|---|---|
| Other Experimental Properties | There are 47 isotopes and isomers of iridium that are recognized(1). Iridium-191and iridium-193 are naturally occuring and are stable(2). Iridium-165 through 190, iridium-192, and iridium-194 through 198 are artificially produced and are radioactive(2). |
| Chemical Classes | Physical/Radiation -> Radionuclides |
COMPUTED DESCRIPTORS
| Molecular Weight | 191.96260 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 0 |
| Rotatable Bond Count | 0 |
| Exact Mass | 191.96260 g/mol |
| Monoisotopic Mass | 191.96260 g/mol |
| Topological Polar Surface Area | 0 Ų |
| Heavy Atom Count | 1 |
| Formal Charge | 0 |
| Complexity | 0 |
| Isotope Atom Count | 1 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Iridium Ir 192 is a radioactive isotope of iridium. Iridium-192 emits gamma rays and has a half-life of 74 days. A high dose rate of this radioisotope can be used in brachytherapy to treat tumors by selectively delivering a cytotoxic dose of radiation to the tumor site.