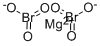CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Magnesium bromate is a crystalline solid. Soluble in water and denser than water. Hence sinks in water. Contact may cause irritation to skin, eyes, and mucous membranes. May be toxic by ingestion, inhalation and skin absorption. Used to make other chemicals. |
|---|
COMPUTED DESCRIPTORS
| Molecular Weight | 280.11 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 0 |
| Exact Mass | 279.78916 g/mol |
| Monoisotopic Mass | 277.79120 g/mol |
| Topological Polar Surface Area | 114 Ų |
| Heavy Atom Count | 9 |
| Formal Charge | 0 |
| Complexity | 36.5 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 3 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Magnesium bromate is a crystalline solid. Soluble in water and denser than water. Hence sinks in water. Contact may cause irritation to skin, eyes, and mucous membranes. May be toxic by ingestion, inhalation and skin absorption. Used to make other chemicals.