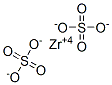CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Anhydrous zirconium sulfate is a colorless microcrystalline solid. Zirconium sulfate is also obtained as a white crystalline tetrahydrate, ZrSO4.4H2O. Both forms are soluble in water and noncombustible. It is corrosive to aluminum. It is used in chemical analysis, as an additive for lubricants, and for many other uses. |
|---|---|
| Color/Form | White crystals |
| Melting Point | 410 °C |
| Solubility | Crystalline solid. Converted to monohydrate at 100 °C, to anhydrous form at 380 °C; solubility in water at 18 °C: 52.5 g/100 g solution. A solution at room temperature deposits a solid on standing. The more dilute the solution the more rapid the decomposition /Zirconium sulfate tetrahydrate/ |
| Density | 3 at 68 °F (approx.) (USCG, 1999) - Denser than water; will sink |
| Stability/Shelf Life | Solution at room temperature deposits a solid on standing. The more dilute the solution, the more rapid the deposition. /Zirconium sulfate tetrahydrate/ |
| Decomposition | When heated to decomposition it emits toxic fumes of /sulfur oxides/. |
| Other Experimental Properties | Hygroscopic |
| Chemical Classes | Metals -> Metals, Inorganic Compounds |
COMPUTED DESCRIPTORS
| Molecular Weight | 283.4 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 0 |
| Exact Mass | 281.808158 g/mol |
| Monoisotopic Mass | 281.808158 g/mol |
| Topological Polar Surface Area | 177 Ų |
| Heavy Atom Count | 11 |
| Formal Charge | 0 |
| Complexity | 62.2 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 3 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Anhydrous zirconium sulfate is a colorless microcrystalline solid. Zirconium sulfate is also obtained as a white crystalline tetrahydrate, ZrSO4.4H2O. Both forms are soluble in water and noncombustible. It is corrosive to aluminum. It is used in chemical analysis, as an additive for lubricants, and for many other uses.