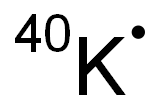CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | A naturally occurring isotope that comprises about 0.012% of total potassium; K-39 (93% of potassium) and K-39 (7% of potassium) are not radioactive. Elemental potassium is a soft, silver-white metal. [Argonne] |
|---|---|
| Chemical Classes | Physical/Radiation -> Radionuclides |
COMPUTED DESCRIPTORS
| Molecular Weight | 39.9639982 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 0 |
| Rotatable Bond Count | 0 |
| Exact Mass | 39.9639982 g/mol |
| Monoisotopic Mass | 39.9639982 g/mol |
| Topological Polar Surface Area | 0 Ų |
| Heavy Atom Count | 1 |
| Formal Charge | 0 |
| Complexity | 0 |
| Isotope Atom Count | 1 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Potassium-40 is a naturally occurring radioactive isotope of potassium. Potassium is the chemical element with the symbol K and atomic number 19. Potassium is widely distributed in nature and is present in all plant and animal tissues. Potassium-40 comprises about 0.012% of naturally occurring potassium. It is the predominant radioactive component in human tissues and in most food. Potassium-40 has a half-life of 1.3 billion years and emits beta and gamma radiation. (L1134, L1851)