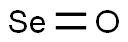CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Selenium dioxide appears as a white or creamy-white volatile lustrous crystal or crystalline powder with a pungent sour smell. Melting point 340 deg C. Density 3.954 g / cm3. Toxic by ingestion and inhalation. |
|---|---|
| Color/Form | Lustrous tetragonal needles; yellowish green vapor |
| Odor | Vapor has a pungent sour smell |
| Taste | Acidic taste; leaves a burning sensation. |
| Boiling Point | 599 °F at 760 mmHg (sublimes) (USCG, 1999) |
| Melting Point | 340 °C under pressure |
| Solubility | Soluble in concentrated sulfuric acid |
| Density | 3.95 at 68 °F (USCG, 1999) - Denser than water; will sink |
| Vapor Pressure | 12.5 mm Hg at 70 °C; 20.2 mm Hg at 94 °C; 39.0 mm Hg at 181 °C; 760 mm Hg at 315 °C; 848 mm Hg at 320 °C |
| Stability/Shelf Life | Stable to light and heat. |
| Decomposition | When heated to decomposition ... emits toxic fumes of /selenium/. |
| Corrosivity | In presence of water will corrode most metals |
| Odor Threshold | 0.0002 mg/cu m |
| Refractive Index | Index of refraction: less than 1.76 at 20 °C/D |
| Other Experimental Properties | Dipole moment: 2.62 debye |
| Chemical Classes | Other Classes -> Other Inorganic Compounds |
COMPUTED DESCRIPTORS
| Molecular Weight | 110.97 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 111.90635 g/mol |
| Monoisotopic Mass | 111.90635 g/mol |
| Topological Polar Surface Area | 34.1 Ų |
| Heavy Atom Count | 3 |
| Formal Charge | 0 |
| Complexity | 18.3 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Selenium dioxide appears as a white or creamy-white volatile lustrous crystal or crystalline powder with a pungent sour smell. Melting point 340 deg C. Density 3.954 g / cm3. Toxic by ingestion and inhalation.