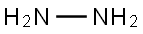| Physical Description |
Hydrazine, anhydrous appears as a colorless, fuming oily liquid with an ammonia-like odor. Flash point 99 °F. Explodes during distillation if traces of air are present. Toxic by inhalation and by skin absorption. Corrosive to tissue. Produces toxic oxides of nitrogen during combustion. Used as a rocket propellant and in fuel cells. |
| Color/Form |
Colorless oily liquid |
| Odor |
Penetrating odor resembling ammonia. |
| Boiling Point |
236.3 °F at 760 mmHg (EPA, 1998) |
| Melting Point |
36 °F (EPA, 1998) |
| Flash Point |
125.6 °F (EPA, 1998) |
| Solubility |
Miscible (NIOSH, 2023) |
| Density |
1.011 at 59 °F (EPA, 1998) - Denser than water; will sink |
| Vapor Density |
Relative vapor density (air = 1): 1.1 |
| Vapor Pressure |
14.4 mmHg at 77 °F (EPA, 1998) |
| LogP |
log Kow = -2.07 |
| Stability/Shelf Life |
Stable under recommended storage conditions. |
| Autoignition Temperature |
518 °F (USCG, 1999) |
| Decomposition |
Hazardous decomposition products formed under fire conditions - Nitrogen oxides (NOx). |
| Viscosity |
0.974 uPa-sec at 20 °C |
| Heat of Combustion |
-8345 btu/Lb = -4636 cal/g = -194.1X10+5 J/kg |
| Heat of Vaporization |
44.7 kJ/mol at 25 °C |
| pH |
Aqueous solutions of hydrazine are highly alkaline. pH of a 64 wt% aqueous solution of hydrazine is 12.75. |
| Surface Tension |
66.7 mN/m at 25 °C |
| Ionization Potential |
8.93 eV |
| Odor Threshold |
Odor Threshold Low: 3.0 [mmHg] Odor Threshold High: 4.0 [mmHg] AIHA detection odor threshold AIHA (mean = 3.7 ppm) |
| Refractive Index |
Surface tension 74.3 mN/m at 25 °C: Viscosity: 1.5 mPa.s at 25 °C: Index of refraction: 1.4644 at 25 °C/D; Heat of formation -242.71 kJ/mol /Hydrazine hydrate/ |
| Dissociation Constants |
pKa = 7.96 |
| Other Experimental Properties |
Forms salts with inorganic acids. Highly polar solvent. Powerful reducing agent. |
| Chemical Classes |
Nitrogen Compounds -> Hydrazines |