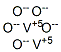CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Vanadium pentoxide appears as a yellow to red crystalline powder. Slightly soluble in water and denser than water. Contact may cause severe irritation to skin, eyes, and mucous membranes. May be toxic by ingestion, inhalation and skin absorption. |
|---|---|
| Color/Form | Yellow to rust-brown orthorhombic crystals |
| Odor | Odorless |
| Boiling Point | 3182 °F at 760 mmHg decomposition (EPA, 1998) |
| Melting Point | 1274 °F (EPA, 1998) |
| Solubility | less than 1 mg/mL at 68 °F (NTP, 1992) |
| Density | 3.357 at 64.4 °F (EPA, 1998) - Denser than water; will sink |
| Vapor Pressure | Approximately 0 at 68F (EPA, 1998) |
| Stability/Shelf Life | Stable under recommended storage conditions. |
| Decomposition | Decomposes at 1750 °C |
| pH | pH = 2.7, saturated aqueous solution at 20 °C |
| Refractive Index | Index of refraction: 1.46, 1.52, 1.76 |
| Other Experimental Properties | Loses oxygen reversibly in the region 700-1125 °C; its acid solutions are reduced by sulfur dioxide, zinc and hydrochloric acid and by evaporation with hydrochloric acid; forms red and yellow solutions in concentrated acids; forms vanadates in alkalis |
| Chemical Classes | Metals -> Metals, Inorganic Compounds |
COMPUTED DESCRIPTORS
| Molecular Weight | 181.88 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 0 |
| Exact Mass | 181.862488 g/mol |
| Monoisotopic Mass | 181.862488 g/mol |
| Topological Polar Surface Area | 77.5 Ų |
| Heavy Atom Count | 7 |
| Formal Charge | 0 |
| Complexity | 124 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Vanadium pentoxide appears as a yellow to red crystalline powder. Slightly soluble in water and denser than water. Contact may cause severe irritation to skin, eyes, and mucous membranes. May be toxic by ingestion, inhalation and skin absorption.