Sodium hydroxide
Synonym(s):NaOH;Sodium hydroxide;Caustic Soda;Sodium hydroxide solution;Sodium Hydrate
- CAS NO.:1310-73-2
- Empirical Formula: NaOH
- Molecular Weight: 39.99711
- MDL number: MFCD00011405
- EINECS: 215-185-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-04-24 19:16:04
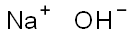
What is Sodium hydroxide?
Absorption
There are no quantitative data for the absorption of sodium hydroxide through the skin. Solutions which contain 50 % sodium hydroxide have been shown to be corrosive and lethal when applied dermally to mice .
Toxicity
Human poisoning cases indicate that a dose of 10 grams orally is fatal . Sodium hydroxide is toxic by oral ingestion .
Sodium hydroxide is corrosive to all tissues. Concentrated vapors lead to serious damage to the eyes and respiratory system. Oral ingestion of sodium hydroxide, which occurs frequently in children, causes severe tissue necrosis, with stricture formation of the esophagus, often resulting in death. Contact with the skin may result in contact dermatitis, hair loss, as well as necrosis due to severe irritation .
Increased incidence of esophageal carcinoma after severe intoxication with sodium hydroxide has been reported in man. In animal studies, long-term dermal contact with substances leading to pH changes in the skin causes the development of tumors, as a result of severe tissue irritation and reparative cell growth .
Mutagenic for mammalian somatic cells. May cause damage to the following organs: mucous membranes, upper respiratory tract, skin, eyes . Tumors are not to be expected if the effects of irritation are prevented .
To date, there are no relevant studies of the prenatal toxic effects of sodium hydroxide .
The Uses of Sodium hydroxide
Sodium hydroxide is one of the most important industrial chemicals. It is used in manufacturing a large number of compounds including several sodium salts, in treating cellulose for producing rayon and cellophane, and in manufacturing soaps, detergents, pulp, and paper. Sodium hydroxide is a common neutralizing agent for acids in acid-base titrations and petroleum refining. Another major application is extracting metals from their ores where alkali fusion, such as fusion with caustic soda, often is applied to open the ores. Additionally, sodium hydroxide is used to precipitate metals as hydroxides. Other uses are in reclaiming rubber, dissolving casein in plastics production, refining vegetable oils, processing textiles, as an eluant in ion chromatography, etching and electroplating, and as a laboratory reagent. Sodium hydroxide also is used as a strong base in many organic synthesis and base-catalyzed reactions. Sodium Hydroxide is a strong base and acid neutralizer also used in the staining procedure of cells.
Background
Sodium hydroxide is also known as lye or soda , or caustic soda . At room temperature, sodium hydroxide is a white crystalline odorless solid that absorbs moisture from the air. It is a synthetically manufactured substance. When dissolved in water or neutralized with acid it releases substantial amounts of heat, which may prove sufficient to ignite combustible materials. Sodium hydroxide is highly corrosive .
Indications
Used to destroy or kill the nail matrix (matrixectomies) .
Pharmacokinetics
Sodium Hydroxide 10% forms a strongly alkaline and caustic solution. As a caustic agent, it is used to destroy organic tissue by chemical action .
Properties of Sodium hydroxide
| Melting point: | 681 °C(lit.) |
| Boiling point: | 1390°C |
| Density | 1.515 g/mL at 20 °C |
| Flash point: | 176-178°C |
| storage temp. | room temp |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| form | beads |
| appearance | White solid |
| color | White |
| Odor | Odorless |
| Water Solubility | SOLUBLE |
| Sensitive | Air Sensitive & Hygroscopic |
Safety information for Sodium hydroxide
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05 |
| GHS Hazard Statements |
H290:Corrosive to Metals H314:Skin corrosion/irritation |
| Precautionary Statement Codes |
P234:Keep only in original container. P260:Do not breathe dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Sodium hydroxide
Abamectin manufacturer
ANJI BIOSCIENCES
SUKHA CHEMICAL INDUSTRIES
Soham Chemical Industries
Nyne Organics Pvt Ltd
UNILOSA INTERNATINAL PRIVATE LIMITED
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran

You may like
-
 Caustic Soda Flakes 99%View Details
Caustic Soda Flakes 99%View Details -
 Caustic Soda Flakes 98%View Details
Caustic Soda Flakes 98%View Details -
 Caustic Soda flakes 99%View Details
Caustic Soda flakes 99%View Details -
 Caustic Soda Flakes 99%View Details
Caustic Soda Flakes 99%View Details -
 Caustic Soda Flakes 99%View Details
Caustic Soda Flakes 99%View Details -
 Sodium hydroxide 99%View Details
Sodium hydroxide 99%View Details -
 Sodium hydroxide 98%View Details
Sodium hydroxide 98%View Details -
 Sodium hydroxide 98%View Details
Sodium hydroxide 98%View Details