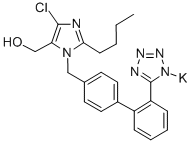
Losartan potassium
Synonym(s):2-butyl-4-chloro-1-[[2′-(2H-tetrazol-5-yl)[1,1′-biphenyl]-4-yl]methyl]- 1H-Imidazole-5-methanol potassium salt;2-Butyl-4-chloro-1-{[2′-(1H-tetrazol-5-yl)(1,1′-biphenyl)-4-yl]methyl}-1H-imidazole-5-methanol monopotassium salt;Losartan potassium;Potassium 5-(4′-((2-butyl-4-chloro-5-(hydroxymethyl)-1H-imidazol-1-yl)methyl)-[1,1′-biphenyl]-2-yl)tetrazol-1-ide
- CAS NO.:124750-99-8
- Empirical Formula: C22H23ClKN6O
- Molecular Weight: 462.01
- MDL number: MFCD02092704
- EINECS: 200-287-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-05-08 14:48:35
What is Losartan potassium?
Description
Losartan potassium is the first potent and selective non-peptide angiotensin II (AT II) AT1 receptor antagonist introduced to the market as a once-daily oral antihypertensive. It is efficacious and long lasting in controlling blood pressure in spontaneous hypertensive rats, in patients with essential hypertension in addition to those patients with renal impairment. Since losartan functions by competitive antagonism at the level of receptor, which represents the most direct way of selectively inhibiting the renin-angiotensin system (RAS) independent of the source of AT II, its use has been reported to be free of the coughing side effect exhibited by the ACE inhibitors. In contrast to calcium channel blockers, losartan does not appear to cause ankle edema, headache and tachycardia. It is also reported to be in clinical trials for the treatment of heart failure. Other reports indicate that losartan may have potential efficacy as an anxiolytic, an antiglaucoma agent, in addition to in providing protection against stroke and in preventing the myointimal proliferative response of the vascular wall after coronary angioplasty and surgery.
The Uses of Losartan potassium
A nonpeptide angiotensin II AT1-receptor antagonist. Antihypertensive.
What are the applications of Application
Losartan Potassium is a specific antagonist of the AT1 receptor with significant downstream effects.
Chemical Properties
Losartan potassium is a white to off-white free-flowing crystalline powder.Oxidation of the 5-hydroxymethyl group on the imidazole ring results in the active metabolite of losartan.
Metabolism
Losartan potassium is an orally active agent that undergoes substantial first-pass metabolism by cytochrome P450 enzymes. It is converted, in part, to an activecarboxylic acid metabolite that is responsible for most of the angiotensin II receptor antagonism that follows losartan treatment. About 14% of an orally-administereddose of losartan is converted to the active metabolite. In addition to the active carboxylic acid metabolite, several inactive metabolites are formed. In vitro studiesindicate that cytochrome P450 2C9 and 3A4 are involved in the biotransformation of losartan to its metabolites.
Side Effects
Side effects of Losartan potassium include:
diarrhea
stomach pain
muscle cramps
leg or back pain
dizziness
headache
sleep problems (insomnia)
tiredness, and
cold or flu symptoms such a stuffy nose, sneezing,sore throat, fever, and cough
Properties of Losartan potassium
| Melting point: | 263-265°C |
| storage temp. | Inert atmosphere,Room Temperature |
| solubility | Freely soluble in water and in methanol, slightly soluble in acetonitrile. |
| form | powder or crystals |
| color | Off-white |
Safety information for Losartan potassium
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P312:Call a POISON CENTER or doctor/physician if you feel unwell. P302+P352:IF ON SKIN: wash with plenty of soap and water. P304+P340:IF INHALED: Remove victim to fresh air and Keep at rest in a position comfortable for breathing. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P403+P233:Store in a well-ventilated place. Keep container tightly closed. |
Computed Descriptors for Losartan potassium
| InChIKey | OXCMYAYHXIHQOA-UHFFFAOYSA-N |
Abamectin manufacturer
TAGOOR LABORATORIES PVT LTD
HEMA PHARMACEUTICALS PVT LTD
Vijayasri Organics Private Limited
AVD pharmaceuticals Pvt Ltd
SGMR PHARMACEUTICALS PVT LTD
Dr. Reddy's Laboratories Ltd
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran


You may like
-
 Losartan Potassium 99%View Details
Losartan Potassium 99%View Details -
 124750-99-8 98%View Details
124750-99-8 98%View Details
124750-99-8 -
 Losartan Potassium 98%View Details
Losartan Potassium 98%View Details
124750-99-8 -
 LOSARTAN POTASSIUM 99%View Details
LOSARTAN POTASSIUM 99%View Details -
 Losartan potassium 99%View Details
Losartan potassium 99%View Details -
 Losartan potassium 98%View Details
Losartan potassium 98%View Details -
 Losartan potassium 98%View Details
Losartan potassium 98%View Details -
 124750-99-8 Losartan potassium 98%View Details
124750-99-8 Losartan potassium 98%View Details
124750-99-8