1,1'-Carbonyldiimidazole
Synonym(s):1,1′-Carbonyldiimidazole;1,1′-Carbonyl-diimidazole;CDI
- CAS NO.:530-62-1
- Empirical Formula: C7H6N4O
- Molecular Weight: 162.15
- MDL number: MFCD00005286
- EINECS: 208-488-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-05-18 08:51:57
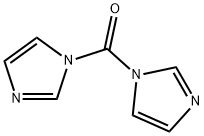
What is 1,1'-Carbonyldiimidazole?
Description
Carbonyl diimidazole (CDI) is a useful alternative to highly toxic phosgene. CDI is similar to phosgene in its synthetic utility but is easier to handle since it exists as a solid. CDI is moisture sensitive and reacts readily with H2O to evolve CO2. CDI can be used for the activation of carboxylic acids. These activated carboxylic acids can be reacted with nucleophiles to make product that such as amides, esters, ureas, and carbamates. CDI reactions often don’t require a base because the liberated imidazole anion acts a base.
The Uses of 1,1'-Carbonyldiimidazole
Peptide coupling reagent1,1'-Carbonyldiimidazole acts as a coupling reagent and utilized for coupling of amino acids in order to prepare peptide in organic synthesis. It is also used in the preparation of beta-keto sulfones, sulfoxides and beta-enamino acid derivatives. It is used to convert alcohols and amines into carbamates, esters, and ureas. It is involved in the preparation of formylized imidazole by reaction with formic acid. Further, it is used in the synthesis of dipolar polyamides compounds. In addition to this, it is considered as an equivalent of phosgene and used to prepare asymmetric bis alkyl carbonate.
The Uses of 1,1'-Carbonyldiimidazole

The acid (152 mg, 0.801 mmol) was added to a solution of CDI (130 mg, 0.801 mmol) in dry THF (1 mL) at RT under N2. The mixture was stirred for 90 min at RT, after which time it was added over 25 min to a solution of the amine (200 mg, 0.400 mmol) in THF (1.5 mL) at RT under N2. The reaction mixture was stirred at RT for 16 h. Additional acid (152 mg, 0.801 mmol) and CDI (130 mg, 0.801 mmol) in dry THF (1 mL) was added slowly at RT. The reaction mixture was stirred at RT for 2 h. TFA (1 mL, 26.1 mmol) was added at RT, and the mixture was stirred 22 h. Additional TFA (1 mL, 26.1 mmol) was added, and the mixture was stirred 18 h. The reaction mixture was then stirred at 80 C for 160 min. The mixture was diluted with H2O and extracted with EtOAc. The org phase was washed with brine, dried (MgSO4), and concentrated to give an oil. The oil was purified by Prep LC (12 g silica, 100% DCM to 90:9:1 DCM/MeOH/NH3) to provide the product as a white solid. [158 mg, 80%]
What are the applications of Application
N,N′-Carbonyldiimidazole is a versatile coupling agent used in the synthesis of dipolar polyamides
Properties of 1,1'-Carbonyldiimidazole
| Melting point: | 117-122 °C(lit.) |
| Boiling point: | 288.83°C (rough estimate) |
| Density | 1.303 g/mL at 25 °C |
| storage temp. | 2-8°C |
| solubility | Soluble in dimethylformamide. |
| form | Crystalline Powder |
| appearance | White cystalline solid |
| color | White to light cream |
| Water Solubility | Insoluble in water |
| Sensitive | Moisture Sensitive |
Safety information for 1,1'-Carbonyldiimidazole
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05  Exclamation Mark Irritant GHS07  Health Hazard GHS08 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H314:Skin corrosion/irritation |
| Precautionary Statement Codes |
P260:Do not breathe dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for 1,1'-Carbonyldiimidazole
| InChIKey | PFKFTWBEEFSNDU-UHFFFAOYSA-N |
Abamectin manufacturer
PURALABS
VGS SYNTHESIS PRIVATE LIMITED
vj finechem pvt ltd
THE MOLECULEZ
SGMR PHARMACEUTICALS PVT LTD
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE) 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THFRelated products of tetrahydrofuran

You may like
-
 CDI 530-62-1 99%View Details
CDI 530-62-1 99%View Details
530-62-1 -
 N, N- Carbonyl diimidazole (CDI) 98%View Details
N, N- Carbonyl diimidazole (CDI) 98%View Details
530-62-1 -
 Carbonyldiimidazole 530-62-1 98%View Details
Carbonyldiimidazole 530-62-1 98%View Details
530-62-1 -
 Carbonyldiimidazole 98%View Details
Carbonyldiimidazole 98%View Details -
 Carbonyldiimidazole 98%View Details
Carbonyldiimidazole 98%View Details -
 Carbonyldiimidazole 98%View Details
Carbonyldiimidazole 98%View Details -
 Carbonyldiimidazole 98%View Details
Carbonyldiimidazole 98%View Details -
 Carbonyldiimidazole 98%View Details
Carbonyldiimidazole 98%View Details